1.2.1
Structure of Carbohydrates
Test your knowledge with free interactive questions on Seneca — used by over 10 million students.
Types of Monosaccharides
Carbohydrates are made of carbon, hydrogen and oxygen atoms. They are made from monosaccharides, which are simple sugars containing three to seven carbon atoms.
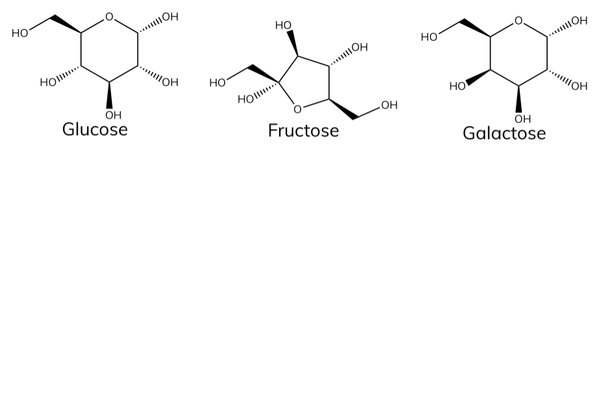
Examples of monosaccharides
- Monosaccharides are simple sugars. Examples include:
- Glucose.
- Galactose (found in milk).
- Fructose (found in fruit).
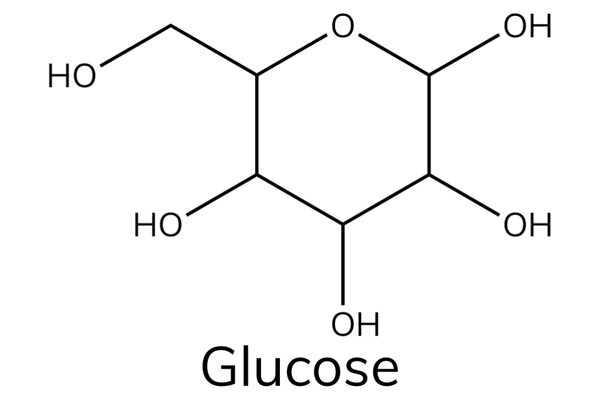
Glucose
- Glucose is a hexose sugar that has the chemical formula C6H12O6.
- Glucose is an important source of energy in humans.
- During cellular respiration, the energy released from glucose helps to make adenosine triphosphate (ATP).
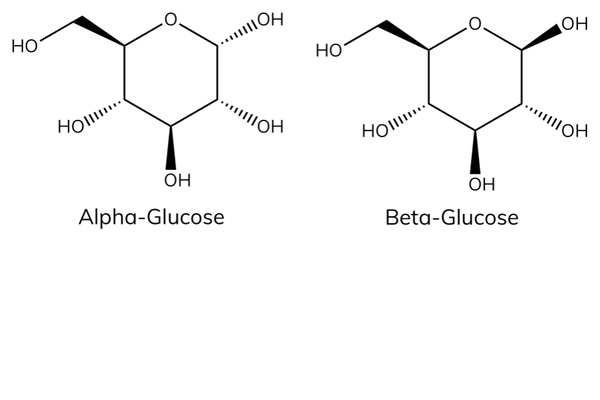
Alpha vs beta glucose
- Alpha- and beta-glucose are isomers. Isomers have the same molecular formula but a different arrangement of atoms in space.
- The carbon atoms are numbered from 1 – 6 and the OH (hydroxyl) groups are in a different orientation around C1.
Disaccharides and Polysaccharides
When two monosaccharides join via a condensation reaction, they form a disaccharide. When more than two disaccharides join together, they form a polysaccharide chain.
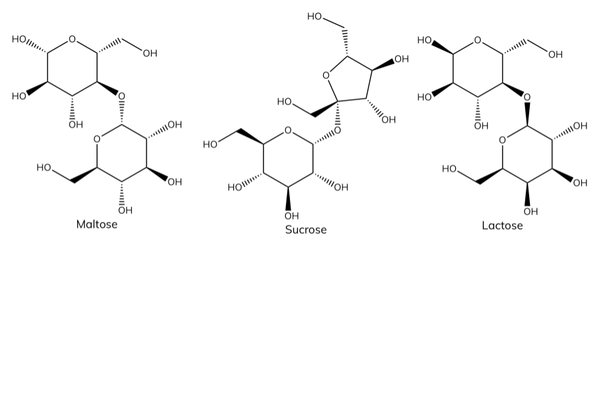
Examples of disaccharides
- Glucose + glucose → maltose.
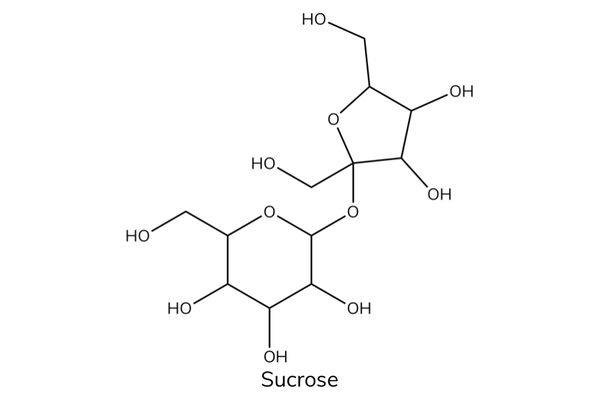
- Glucose + fructose → sucrose.
- Glucose + galactose → lactose.
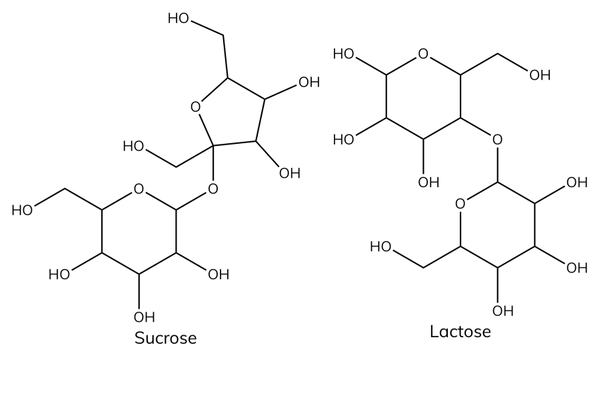
Functions of disaccharides
- Sucrose is common table sugar.
- Lactose is the sugar found in milk.
- Lactose intolerance is a common problem where the body is unable to digest lactose.
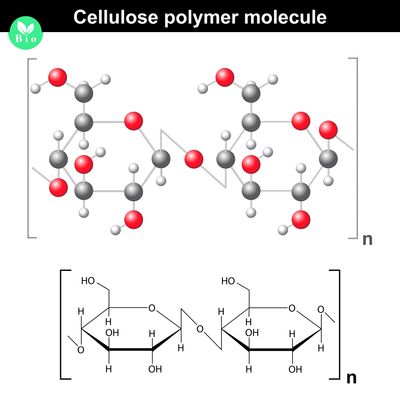
Polysaccharides
- Polysaccharides are made up of three or more monosaccharides joined together by glycosidic bonds.
- The chain may be branched or unbranched.
- The chain may contain different types of monosaccharides.
- Starch, glycogen, cellulose and chitin are examples of polysaccharides.
Benedict's Test for Sugars
Benedict’s solution (also known as Benedict's reagent or the Benedict’s test) can be used as a test for reducing and non-reducing sugars.

Reducing sugars
- All monosaccharides are reducing sugars.
- E.g. Glucose, galactose and fructose.
- Some disaccharides are reducing sugars.
- E.g. Lactose and maltose.
Test for reducing sugars
- Benedict's solution can be reduced by reducing sugars.
- Benedict's solution is a clear blue liquid that changes colour and gives a precipitate depending on how much it is reduced.
- Step 1: Place 2 ml of the substance in a boiling tube (substance must be in liquid form).
- Step 2: Add 10 drops of Benedict's solution.
- Step 3: Place in a boiling water bath for 3-5 minutes.

Results of the Benedict's test
- Blue solution → no reducing sugar.
- Green/yellow precipitate → traces of reducing sugar.
- Orange/red precipitate → moderate amounts of reducing sugar.
- Brick red precipitate → large amount of reducing sugar.
Non-reducing sugars
- Non-reducing sugars will show a negative result to the Benedict’s test. A second test is needed to determine if non-reducing sugar is present.
- Sucrose is a non-reducing sugar. It is a disaccharide made up of glucose and fructose joined by a glycosidic bond.

Test for non-reducing sugars
- Step 1: Boil in dilute HCl (to hydrolyse the non-reducing sugar).
- Step 2: Neutralise the solution by adding sodium hydrogen carbonate.
- Step 3: Repeat the Benedict’s test.
- The result will now be positive if a non-reducing sugar is present.
- If the solution remains blue, then no sugar is present.
1Biological Molecules
1.1Monomers & Polymers
1.2Carbohydrates
1.3Lipids
1.4Proteins
1.5Nucleic Acids
1.6ATP
1.7Water
1.8Inorganic Ions
2Cells
2.1Cell Structure
2.2Mitosis & Cancer
2.3Transport Across Cell Membrane
2.4Cell Recognition & the Immune System
3Substance Exchange
3.1Surface Area to Volume Ratio
3.2Gas Exchange
3.3Digestion & Absorption
3.4Mass Transport
4Genetic Information & Variation
4.1DNA, Genes & Chromosomes
4.2DNA & Protein Synthesis
4.3Mutations & Meiosis
4.4Genetic Diversity & Adaptation
4.5Species & Taxonomy
4.6Biodiversity Within a Community
4.7Investigating Diversity
5Energy Transfers (A2 only)
5.1Photosynthesis
5.2Respiration
5.3Energy & Ecosystems
6Responding to Change (A2 only)
6.1Nervous Communication
6.2Nervous Coordination
6.3Muscle Contraction
6.4Homeostasis
7Genetics & Ecosystems (A2 only)
7.1Genetics
7.2Populations
7.3Evolution
8The Control of Gene Expression (A2 only)
8.1Mutation
8.2Gene Expression
8.3Genome Projects
Jump to other topics
1Biological Molecules
1.1Monomers & Polymers
1.2Carbohydrates
1.3Lipids
1.4Proteins
1.5Nucleic Acids
1.6ATP
1.7Water
1.8Inorganic Ions
2Cells
2.1Cell Structure
2.2Mitosis & Cancer
2.3Transport Across Cell Membrane
2.4Cell Recognition & the Immune System
3Substance Exchange
3.1Surface Area to Volume Ratio
3.2Gas Exchange
3.3Digestion & Absorption
3.4Mass Transport
4Genetic Information & Variation
4.1DNA, Genes & Chromosomes
4.2DNA & Protein Synthesis
4.3Mutations & Meiosis
4.4Genetic Diversity & Adaptation
4.5Species & Taxonomy
4.6Biodiversity Within a Community
4.7Investigating Diversity
5Energy Transfers (A2 only)
5.1Photosynthesis
5.2Respiration
5.3Energy & Ecosystems
6Responding to Change (A2 only)
6.1Nervous Communication
6.2Nervous Coordination
6.3Muscle Contraction
6.4Homeostasis
7Genetics & Ecosystems (A2 only)
7.1Genetics
7.2Populations
7.3Evolution
8The Control of Gene Expression (A2 only)
8.1Mutation
8.2Gene Expression
8.3Genome Projects
Practice questions on Structure of Carbohydrates
Can you answer these? Test yourself with free interactive practice on Seneca — used by over 10 million students.
- 1What is the chemical formula for glucose?Multiple choice
- 2What are the products of the hydrolysis of sucrose?Multiple choice
- 3
- 4What can the Benedict’s test be used to test?Fill in the list
- 5Test for Non-Reducing SugarsPut in order
Unlock your full potential with Seneca Premium
Unlimited access to 10,000+ open-ended exam questions
Mini-mock exams based on your study history
Unlock 800+ premium courses & e-books