2.5.5
Reactions of Alkenes
Test your knowledge with free interactive questions on Seneca — used by over 10 million students.
Alkenes
Alkenes are molecules with a double covalent bond. There is a high electron density between the carbon atoms because of the double bond.

Reactions of alkenes
- Alkenes react with several reagents in addition reactions.
- The alkenes are nucleophiles because of the high electron density between the carbon atoms.
- The mechanism is shown next for the reaction with hydrogen bromide.
- The mechanism is called the electrophilic addition mechanism (because an electrophile is added).
- They will react in a similar way with sulfuric acid, and bromine.

Electrophilic addition
- Above shows the reaction with HBr.

With bromine
- The reaction can also be done with Br2.
- The partial charges are assigned to the bromine atoms because the high electron density of the double bond repels the bonding electrons and polarises the bond - this is shown in the mechanism.
Uses of Electrophilic Addition
Electrophilic addition reactions between alkenes and bromine as well as alkenes and sulfuric acid are useful.

With bromine
- The reaction with bromine is used as a test for alkenes.
- Bromine water is a light orange colour and will be decolourised in the presence of an alkene (light orange → colourless).
- This test works because bromine can add to alkenes via an electrophilic addition reaction.

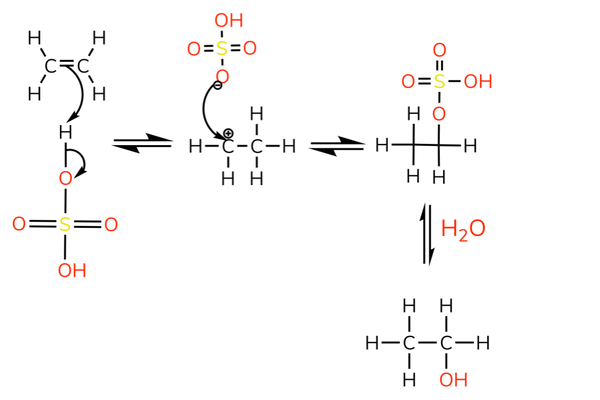
With sulfuric acid
- The reaction with sulfuric acid is used to produce alcohols.
- The sulfate ion is removed by nucleophilic substitution.
Asymmetric Electrophilic Addition
Asymmetric alkenes can have multiple products in electrophilic addition reactions.
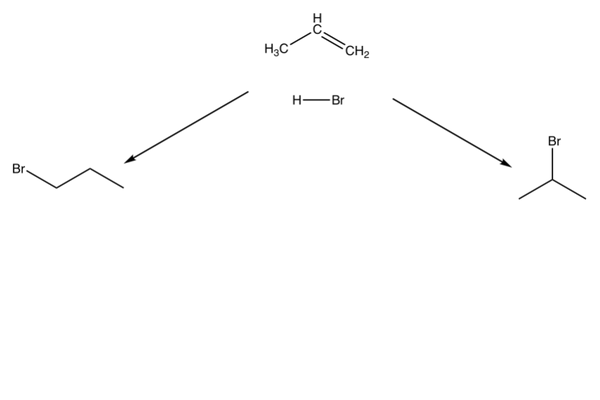
Multiple products
- If you have an asymmetric alkene, you can get multiple products.
- A reaction will favour one of the products over the other - we call this selectivity.
- This is shown for the reaction of propene with HBr.
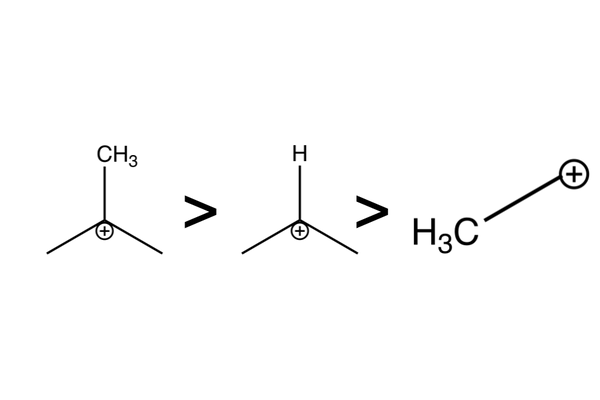
Intermediate stability
- This selectivity is driven by carbocation intermediate stability:
- Tertiary carbocations > secondary carbocations > primary carbocations.
- The more alkyl groups there are next to the positive charge, the more stable the intermediate is.
- Alkyl groups push electrons and so help to stabilise adjacent positive charge.

Which product when?
- When considering which product is most likely, we must consider the intermediates formed.
- 2-bromopropane is the most likely product because the intermediate is a secondary carbocation, rather than a primary carbocation.
- This is because the secondary carbocation is more stable.
- It has more alkyl groups pushing electrons onto the positive charge.
- This is because the secondary carbocation is more stable.
- Both reactions will happen and you will get both products, but you will have one major and one minor.
1Structure - Models of the Particulate of Matter
1.1Introduction to the Particulate Model of Matter
1.2The Nuclear Atom
1.3Electron Configuration
1.4Counting Particles by Mass: The Mole
1.6Elements, Compounds & Mixtures
1.7States of Matter & Changes of State
1.8Reacting Masses &. Volumes
1.9Solutions
2Structure - Models of Bonding & Structure
2.1The Ionic Model
2.2The Covalent Model
2.3Covalent Structures
2.4The Metallic Model
2.5From Models to Materials
2.6Valence Electrons & Ionic Compounds
2.7Molecular Shape
3Structure - Classification of Matter
3.1The Periodic Table: Classification of Elements
3.2Periodic Trends
3.3Group 1 Alkali Metals
3.4Halogens
3.5Noble gases, group 18
3.6Functional Groups: Classification of Organic
3.7Functional Group Chemistry
3.8Alkanes
3.9Alcohols
4Reactivity - What Drives Chemical Reaction?
4.1Endothermic & Exothermic Reactions
4.2Enthalpy of Reaction, Formation, & Hess' Law
5Reactivity - How Much, How Fast & How Far?
5.1Kinetics
5.2Rates of Reaction
5.3Stoichometry
5.4Le Châtelier’s Principle
5.5Introduction to Equilibrium
5.6Equilibrium Constant
5.7Reaction Quotient & Equilibrium Constant
6Reactivity - The Mechanisms of Chemical Change
6.1Proton Transfer Reactions
6.2The pH Scale
6.3Strong & Weak Acids and Bases
6.4Acid Deposition
6.5Types of Organic Reactions
6.6Oxidation & Reduction
6.7Electrochemical Cells
6.9Acid-Base Titrations
6.9.1Titration Calculation Weak Acid & Strong Base6.9.2Titration Experimental Detail6.9.3Extended Response - Titration6.9.4Titration Calculations6.9.5Titration Curves6.9.6Titration Calculation Strong Acid & Weak Base6.9.7IB Multiple Choice - Titrations6.9.8Polyprotic Acids6.9.9Titration Calculations Strong Acid & Strong Base6.9.10Titrations Curves 2
7Measurement, Data Processing & Analysis
7.1Uncertainties & Errors in Measurements & Results
7.2Graphical Techniques
7.3Spectroscopic Identification of Organic Compounds
7.4Infrared Spectroscpy
Jump to other topics
1Structure - Models of the Particulate of Matter
1.1Introduction to the Particulate Model of Matter
1.2The Nuclear Atom
1.3Electron Configuration
1.4Counting Particles by Mass: The Mole
1.6Elements, Compounds & Mixtures
1.7States of Matter & Changes of State
1.8Reacting Masses &. Volumes
1.9Solutions
2Structure - Models of Bonding & Structure
2.1The Ionic Model
2.2The Covalent Model
2.3Covalent Structures
2.4The Metallic Model
2.5From Models to Materials
2.6Valence Electrons & Ionic Compounds
2.7Molecular Shape
3Structure - Classification of Matter
3.1The Periodic Table: Classification of Elements
3.2Periodic Trends
3.3Group 1 Alkali Metals
3.4Halogens
3.5Noble gases, group 18
3.6Functional Groups: Classification of Organic
3.7Functional Group Chemistry
3.8Alkanes
3.9Alcohols
4Reactivity - What Drives Chemical Reaction?
4.1Endothermic & Exothermic Reactions
4.2Enthalpy of Reaction, Formation, & Hess' Law
5Reactivity - How Much, How Fast & How Far?
5.1Kinetics
5.2Rates of Reaction
5.3Stoichometry
5.4Le Châtelier’s Principle
5.5Introduction to Equilibrium
5.6Equilibrium Constant
5.7Reaction Quotient & Equilibrium Constant
6Reactivity - The Mechanisms of Chemical Change
6.1Proton Transfer Reactions
6.2The pH Scale
6.3Strong & Weak Acids and Bases
6.4Acid Deposition
6.5Types of Organic Reactions
6.6Oxidation & Reduction
6.7Electrochemical Cells
6.9Acid-Base Titrations
6.9.1Titration Calculation Weak Acid & Strong Base6.9.2Titration Experimental Detail6.9.3Extended Response - Titration6.9.4Titration Calculations6.9.5Titration Curves6.9.6Titration Calculation Strong Acid & Weak Base6.9.7IB Multiple Choice - Titrations6.9.8Polyprotic Acids6.9.9Titration Calculations Strong Acid & Strong Base6.9.10Titrations Curves 2
7Measurement, Data Processing & Analysis
7.1Uncertainties & Errors in Measurements & Results
7.2Graphical Techniques
7.3Spectroscopic Identification of Organic Compounds
7.4Infrared Spectroscpy
Practice questions on Reactions of Alkenes
Can you answer these? Test yourself with free interactive practice on Seneca — used by over 10 million students.
- 1What is a typical reaction for alkenes?Multiple choice
- 2Which of the following is true for alkenes?True / false
- 3What must be reacted together to give dibromopropane?Fill in the list
- 4
- 5
Unlock your full potential with Seneca Premium
Unlimited access to 10,000+ open-ended exam questions
Mini-mock exams based on your study history
Unlock 800+ premium courses & e-books