2.2.6
The pH Scale
Test your knowledge with free interactive questions on Seneca — used by over 10 million students.
The pH scale
Acids and alkalis all have a pH, which is a number that tells you how strong the acid or alkali is. It starts at zero and goes up to 14.
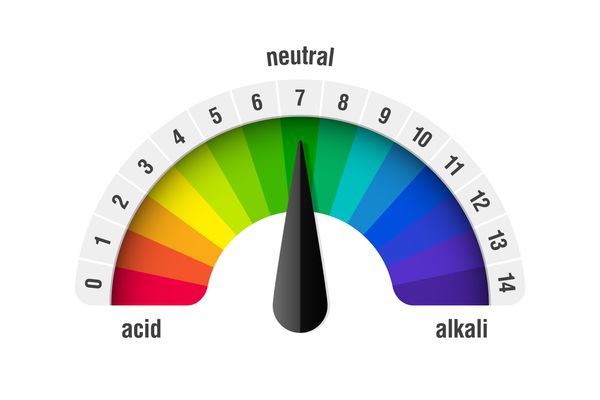
The pH scale
- Anything below 7 is an acid.
- The lower the number, the stronger the acid.
- Anything above 7 is an alkali.
- The higher the number, the stronger the alkali. pH 7 is neutral – these chemicals are neither acid or alkali.
- Pure water has a pH of 7.
- Each pH number is also given a colour.
- The colours on the pH scale are what colour the chemical will go if ‘universal indicator’ is added.

Common acids
- Lemon juice.
- Hydrochloric acid.
- Sulfuric acid.
- Nitric acid.
- Vinegar.

Common alkalis
- Soap.
- Toothpaste.
- Ammonia.
- Sodium hydroxide.
- Lithium hydroxide.
- Calcium oxide.
1Biology
1.1Cells, Tissues & Organs
1.1.1Microscopes1.1.2Magnification1.1.3Multicellular Organisms1.1.4Tissues1.1.5Organs1.1.6Unicellular Organisms1.1.7Diffusion1.1.8Factors Affecting Diffusion1.1.9Plant Cells1.1.10Cellulose1.1.11Plant Tissues1.1.12Leaves1.1.13Animal Cells1.1.14Comparing Animal & Plant Cells1.1.15How to Make a Model Animal and Plant Cell1.1.16Specialised Cells1.1.17Stem Cells1.1.18Uses of Stem Cells1.1.19Disadvantages of Stem Cells1.1.20Blood Components1.1.21Platelets1.1.22The Lungs1.1.23Breathing1.1.24Plant Gas Exchange1.1.25Drugs1.1.26Diagnostic Misconceptions - Microbes1.1.27Diagnostic Misconceptions - Diff. vs Specialised1.1.28Diagnostic Misconceptions - Types of Cell1.1.29Diagnostic Misconceptions - Plants & Animal Cells1.1.30End of Topic Test - Magnification & Cells1.1.31End of Topic Test - Cells & Organisation1.1.32End of Topic Test - Living Organisms
1.2Reproduction & Variation
1.2.1Reproduction in Humans1.2.2Male Reproductive System1.2.3Female Reproductive System1.2.4Gestation1.2.5Pregnancy1.2.6Puberty1.2.7The Menstrual Cycle1.2.8Reproduction in Plants1.2.9Asexual Reproduction1.2.10Advantages of Asexual Reproduction1.2.11Adaptations1.2.12Evolution1.2.13Pollination1.2.14Dispersal Method1.2.15Variation1.2.16Causes of Variation1.2.17Inheritance1.2.18Species & Selective Breeding1.2.19Genetic Conditions - Cystic Fibrosis1.2.20Genetic Conditions - Haemophilia1.2.21End of Topic Test - Reproduction & Puberty1.2.22End of Topic Test - Reproduction & Variation1.2.23Diagnostic Misconceptions - Asexual
1.3Ecological Relationships & Classification
1.3.1Species Interdependence1.3.2Food Chains & Webs1.3.3Changes to Food Webs - Plants1.3.4Changes to Food Webs - Animals1.3.5Changes to Food Webs - Factors1.3.6Relationships in an Ecosystem1.3.7The Impact of Environmental Change1.3.8Decomposers1.3.9Decay1.3.10Assessing Ecosystems1.3.11Ecological Sampling - Quadrats1.3.12Ecological Sampling1.3.13Required Practical - Estimating Population Size1.3.14Pyramids of Number and Biomass1.3.15Classification of Living Organisms1.3.16Competition Between Organisms1.3.17Adaptations of Plants1.3.18Natural Selection1.3.19Evidence for Evolution1.3.20Environmental Changes & Extinctions1.3.21The Importance of Biodiversity1.3.22Bioaccumulation1.3.23Bioaccumulation - DDT and Mercury1.3.24End of Topic Test - Food Webs1.3.25End of Topic Test - Material Cycles & Energy1.3.26End of Topic Test - Population & Evolution
1.4Digestion & Nutrition
1.4.1Balanced Diets1.4.2Vitamins & Minerals1.4.3Protein1.4.4Lipids, Oils and Fats1.4.5Carbohydrates1.4.6Starch1.4.7Energy Needs1.4.8Dietary Fibre1.4.9Diseases Caused by Nutritional Deficiencies1.4.10Digestion1.4.11Enzymes in Digestion1.4.12Required Practical - Enzymes in Digestion1.4.13Plant Nutrition1.4.14Diagnostic Misconceptions - Digestion1.4.15Diagnostic Misconceptions - Excretion vs Egestion1.4.16End of Topic Test - Nutrition
1.5Plants & Photosynthesis
1.5.1Roots1.5.2Photosynthesis1.5.3Leaves in Photosynthesis1.5.4Rate of Photosynthesis1.5.5Testing the Rate of Photosynthesis1.5.6Water Transport in Plants1.5.7Translocation1.5.8The Carbon Cycle1.5.9Human Activities & Carbon Dioxide1.5.10Diagnostic Misconceptions - Plant Food1.5.11End of Topic Test - Plants & Photosynthesis1.5.12Diagnostic Misconceptions - Transpiration
1.6Biological Systems & Processes
1.6.1Living Organisms1.6.2Dichotomous Keys1.6.3Biomechanics1.6.4Muscles1.6.5The Skeleton1.6.6Bones1.6.7Tendons and Ligaments1.6.8Joints1.6.9Carpals and Tarsals1.6.10Measuring Forces1.6.11Antagonistic Muscle Pairings1.6.12The Respiratory System1.6.13Gas Exchange1.6.14Structure & Function of the Gas Exchange System1.6.15Breathing1.6.16Respiration1.6.17Respiration During Exercise1.6.18Anaerobic Respiration1.6.19Lactic Acid1.6.20Effects of Smoking on the Respiratory System1.6.21Balanced Diets1.6.22Human Growth & Development1.6.23DNA1.6.24Alleles1.6.25Genotype vs Phenotype1.6.26Punnett Squares1.6.27The Double Circulatory System1.6.28Heart and Blood1.6.29Blood Vessels1.6.30The Renal System1.6.31Glucose & Diabetes1.6.32Blood Glucose1.6.33Human Illnesses1.6.34Bacterial and Viral Infections1.6.35Antibiotics1.6.36Vaccinations1.6.37How Antibiotics and Vaccines Work1.6.38The Effects of Recreational Drug Use1.6.39Mental Health1.6.40Depression and Anxiety1.6.41End of Topic Test - Muscles & Bones1.6.42End of Topic Test - Respiration1.6.43End of Topic Test - DNA & The Circulatory System1.6.44End of Topic Test - Human Illnesses & Vaccination
2Chemistry
2.1Particles
2.1.1Particles2.1.2States of Matter2.1.3Compression of Substances2.1.4Shape of Substances2.1.5Changes of State2.1.6Elements and Atoms2.1.7Compounds2.1.8Diffusion2.1.9Changing State2.1.10Pressure2.1.11Temperature Increase in a Gas2.1.12Conservation of Mass2.1.13Purity of Substances2.1.14Pure Substances2.1.15Evaporation2.1.16Mixtures2.1.17Separating Mixtures2.1.18Distillation2.1.19Chromatography2.1.20Solubility2.1.21Investigating Solubility2.1.22Chemical Symbols2.1.23Chemical Formulae2.1.24Polymers2.1.25Ceramics2.1.26Composites2.1.27End of Topic Testing - Substances2.1.28End of Topic Testing - Mixtures2.1.29End of Topic Testing - Chemical Symbols & Polymers2.1.30Diagnostic Misconceptions - Particles2.1.31Diagnostic Misconceptions - Temperature2.1.32Diagnostic Misconceptions - Boiling Liquids
2.2Chemical Reactions
2.3Atoms, Elements, Compounds
2.4The Periodic Table
2.5Materials & the Earth
2.5.1The Composition of The Earth2.5.2The Structure of the Earth2.5.3Igneous Rocks2.5.4Sedimentary Rocks2.5.5Metamorphic Rocks2.5.6The Rock Cycle2.5.7Physical Weathering2.5.8Chemical Weathering2.5.9Biological Weathering2.5.10The Formation of Fossils2.5.11Crude Oil2.5.12The Earth's Early Atmosphere2.5.13The Earth's Atmosphere Today2.5.14Oxygen in the Atmosphere2.5.15Carbon Dioxide in the Atmosphere2.5.16Greenhouse Gases2.5.17Climate Change2.5.18Resources2.5.19Recycling2.5.20Ceramics2.5.21Polymers2.5.22Composites2.5.23End of Topic Test - Earth2.5.24End of Topic Test - Materials2.5.25End of Topic Test - Polymers2.5.26Diagnostic Misconceptions - Air2.5.27Diagnostic Misconceptions - Greenhouse Effect
2.6Reactivity
2.6.1Ions2.6.2Ionic Bonding2.6.3State Symbols2.6.4Balancing Chemical Equations2.6.5Relative Formula Mass2.6.6Calculating the Relative Formula Mass2.6.7The Reactivity Series2.6.8Carbon & The Reactivity Series2.6.9Displacement Reactions2.6.10Displacement Reactions - Halogens2.6.11Alloys2.6.12Metal Alloys2.6.13End of Topic Test - Ions2.6.14End of Topic Test - Reactivity
2.7Energetics
3Physics
3.1Energy
3.1.1Energy Stores & Pathways3.1.2Energy Transfers3.1.3Common Energy Transfers3.1.4Wasted Energy3.1.5Efficiency of Energy Transfer3.1.6Sankey Diagrams3.1.7Heat & Temperature3.1.8Heat Transfer3.1.9Conductors vs Insulators3.1.10Reducing Energy Transfers3.1.11Energy & Power3.1.12Energy in Food3.1.13Calories3.1.14Food Labels3.1.15Power Ratings of Appliances3.1.16Comparing Bulbs3.1.17Fuel Bills3.1.18Calculating Fuel Bills3.1.19Non-Renewable Energy - Fossil Fuels3.1.20Other Non-Renewables3.1.21Renewable Energy - Air & Ground3.1.22Renewable Energy - Water3.1.23Conservation of Energy3.1.24End of Topic Test - Energy3.1.25End of Topic Test - Energy & Non-Renewables3.1.26End of Topic Test - Energy in Things3.1.27Diagnostic Misconceptions - Stationary Objects
3.2Forces & Motion
3.2.1Forces3.2.2Contact Forces3.2.3Balanced Forces3.2.4Force Diagrams & Resultant Forces3.2.5Free Body Diagram - Uses3.2.6Force & Acceleration3.2.7Gravity3.2.8Weight3.2.9Pressure3.2.10Speed3.2.11Relative Motion3.2.12Friction 13.2.13Friction 23.2.14Turning Effect3.2.15Moment3.2.16Equation for Moments3.2.17Water & Air Resistance3.2.18Distance-Time Graphs3.2.19Levers3.2.20Work & Machines3.2.21Work3.2.22Machines3.2.23Elasticity3.2.24Elasticity - Hooke's Law3.2.25Archimedes Principle3.2.26Floating & Sinking3.2.27Vacuums3.2.28Thermal Energy & Conduction3.2.29Convection & Radiation3.2.30Evaporation3.2.31End of Topic Test - Forces3.2.32End of Topic Test - Motion3.2.33End of Topic Test - Work3.2.34End of Topic Test - Vacuums3.2.35Diagnostic Misconceptions - Weight vs Mass3.2.36Diagnostic Misconceptions - Gravity
3.3Waves
3.3.1Waves3.3.2Types of Waves3.3.3Observing Waves3.3.4Wave Speed3.3.5Earthquakes3.3.6Hearing3.3.7Observing Sound3.3.8Sound waves are Longitudinal Waves3.3.9Uses of Sound Waves3.3.10The Interactions of Sound with Different Mediums3.3.11Reflecting Sounds3.3.12The Speed of Sound3.3.13Measuring the Speed of Sound3.3.14The Hearing Range of Humans3.3.15The Human Ear3.3.16Light Waves3.3.17Coloured Light3.3.18Observing Light3.3.19Reflection3.3.20Drawing a Reflected Image3.3.21Refraction of Light 13.3.22Refraction of Light 23.3.23The Human Eye3.3.24The Eye as a Pinhole Camera3.3.25Lenses3.3.26Colour3.3.27Seeing Colour3.3.28Colours of Light3.3.29Drawing Waves3.3.30Wave Interactions3.3.31Comparing Sound & Light3.3.32General Principles of Radiation3.3.33Temperature & Black Bodies3.3.34Production of Electromagnetic Waves3.3.35Sound as Radiation3.3.36Light as Radiation3.3.37End of Topic Test - Sound3.3.38End of Topic Test - Light3.3.39End of Topic Test - Waves3.3.40End of Topic Test - Waves 23.3.41Diagnostic Misconceptions - Reflection3.3.42Diagnostic Misconceptions - Wave Speed
3.4Electricity & Magnetism
3.4.1Circuit Symbols3.4.2Resistors & Diodes3.4.3Electric Current3.4.4Measuring Current3.4.5Potential Difference3.4.6Series Circuits3.4.7Parallel Circuits3.4.8Resistance3.4.9Charges3.4.10Static Electricity3.4.11Magnets3.4.12Magnetic Fields3.4.13The Earth's Field3.4.14Electromagnetism3.4.15Uses of Electromagnets3.4.16Strength of Magnetic Fields3.4.17Circuit Symbols HyperLearning3.4.18End of Topic Test - Electricity3.4.19End of Topic Test - Magnetism3.4.20Diagnostic Misconceptions - Positive Charge3.4.21Diagnostic Misconceptions - Powering a Circuit
3.5Matter
3.5.1Physical Reactions3.5.2Changes of State3.5.3Particles3.5.4Density3.5.5Density & the Particle Model3.5.6The Equation for Density3.5.7Dissolving3.5.8Brownian Motion3.5.9Diffusion3.5.10Filtration3.5.11Solids3.5.12Liquids3.5.13Gases3.5.14Weight & Mass3.5.15Gravity3.5.16Gravitational Field Strength3.5.17Gravity in Space3.5.18Atmospheric Pressure3.5.19Liquid Pressure3.5.20End of Topic Test - States of Matter3.5.21End of Topic Test - Weight3.5.22Diagnostic Misconceptions - Floating
4Disciplinary Knowledge
4.1Thinking Scientifically
4.1.1Strengths & Limitations of Models4.1.2Symbols & Formulae to Represent Scientific Ideas4.1.3Analogies in Science4.1.4Changing Models – Atomic Theory4.1.5Working Safely in the Lab4.1.6Variables4.1.7Variables - Common Experiments4.1.8Writing a Hypothesis & Prediction4.1.9Planning an Experiment4.1.10Maths Skills for Science - Mean4.1.11Maths Skills for Science - Percentage & Equations4.1.12Drawing Scientific Apparatus4.1.13Observation & Measurement Skills4.1.14Types of Data4.1.15Graphs & Charts4.1.16Bias in Science4.1.17Conclude & Evaluate4.1.18End of Topic Test - Scientific Thinking4.1.19End of Topic Test - Experimenting
Jump to other topics
1Biology
1.1Cells, Tissues & Organs
1.1.1Microscopes1.1.2Magnification1.1.3Multicellular Organisms1.1.4Tissues1.1.5Organs1.1.6Unicellular Organisms1.1.7Diffusion1.1.8Factors Affecting Diffusion1.1.9Plant Cells1.1.10Cellulose1.1.11Plant Tissues1.1.12Leaves1.1.13Animal Cells1.1.14Comparing Animal & Plant Cells1.1.15How to Make a Model Animal and Plant Cell1.1.16Specialised Cells1.1.17Stem Cells1.1.18Uses of Stem Cells1.1.19Disadvantages of Stem Cells1.1.20Blood Components1.1.21Platelets1.1.22The Lungs1.1.23Breathing1.1.24Plant Gas Exchange1.1.25Drugs1.1.26Diagnostic Misconceptions - Microbes1.1.27Diagnostic Misconceptions - Diff. vs Specialised1.1.28Diagnostic Misconceptions - Types of Cell1.1.29Diagnostic Misconceptions - Plants & Animal Cells1.1.30End of Topic Test - Magnification & Cells1.1.31End of Topic Test - Cells & Organisation1.1.32End of Topic Test - Living Organisms
1.2Reproduction & Variation
1.2.1Reproduction in Humans1.2.2Male Reproductive System1.2.3Female Reproductive System1.2.4Gestation1.2.5Pregnancy1.2.6Puberty1.2.7The Menstrual Cycle1.2.8Reproduction in Plants1.2.9Asexual Reproduction1.2.10Advantages of Asexual Reproduction1.2.11Adaptations1.2.12Evolution1.2.13Pollination1.2.14Dispersal Method1.2.15Variation1.2.16Causes of Variation1.2.17Inheritance1.2.18Species & Selective Breeding1.2.19Genetic Conditions - Cystic Fibrosis1.2.20Genetic Conditions - Haemophilia1.2.21End of Topic Test - Reproduction & Puberty1.2.22End of Topic Test - Reproduction & Variation1.2.23Diagnostic Misconceptions - Asexual
1.3Ecological Relationships & Classification
1.3.1Species Interdependence1.3.2Food Chains & Webs1.3.3Changes to Food Webs - Plants1.3.4Changes to Food Webs - Animals1.3.5Changes to Food Webs - Factors1.3.6Relationships in an Ecosystem1.3.7The Impact of Environmental Change1.3.8Decomposers1.3.9Decay1.3.10Assessing Ecosystems1.3.11Ecological Sampling - Quadrats1.3.12Ecological Sampling1.3.13Required Practical - Estimating Population Size1.3.14Pyramids of Number and Biomass1.3.15Classification of Living Organisms1.3.16Competition Between Organisms1.3.17Adaptations of Plants1.3.18Natural Selection1.3.19Evidence for Evolution1.3.20Environmental Changes & Extinctions1.3.21The Importance of Biodiversity1.3.22Bioaccumulation1.3.23Bioaccumulation - DDT and Mercury1.3.24End of Topic Test - Food Webs1.3.25End of Topic Test - Material Cycles & Energy1.3.26End of Topic Test - Population & Evolution
1.4Digestion & Nutrition
1.4.1Balanced Diets1.4.2Vitamins & Minerals1.4.3Protein1.4.4Lipids, Oils and Fats1.4.5Carbohydrates1.4.6Starch1.4.7Energy Needs1.4.8Dietary Fibre1.4.9Diseases Caused by Nutritional Deficiencies1.4.10Digestion1.4.11Enzymes in Digestion1.4.12Required Practical - Enzymes in Digestion1.4.13Plant Nutrition1.4.14Diagnostic Misconceptions - Digestion1.4.15Diagnostic Misconceptions - Excretion vs Egestion1.4.16End of Topic Test - Nutrition
1.5Plants & Photosynthesis
1.5.1Roots1.5.2Photosynthesis1.5.3Leaves in Photosynthesis1.5.4Rate of Photosynthesis1.5.5Testing the Rate of Photosynthesis1.5.6Water Transport in Plants1.5.7Translocation1.5.8The Carbon Cycle1.5.9Human Activities & Carbon Dioxide1.5.10Diagnostic Misconceptions - Plant Food1.5.11End of Topic Test - Plants & Photosynthesis1.5.12Diagnostic Misconceptions - Transpiration
1.6Biological Systems & Processes
1.6.1Living Organisms1.6.2Dichotomous Keys1.6.3Biomechanics1.6.4Muscles1.6.5The Skeleton1.6.6Bones1.6.7Tendons and Ligaments1.6.8Joints1.6.9Carpals and Tarsals1.6.10Measuring Forces1.6.11Antagonistic Muscle Pairings1.6.12The Respiratory System1.6.13Gas Exchange1.6.14Structure & Function of the Gas Exchange System1.6.15Breathing1.6.16Respiration1.6.17Respiration During Exercise1.6.18Anaerobic Respiration1.6.19Lactic Acid1.6.20Effects of Smoking on the Respiratory System1.6.21Balanced Diets1.6.22Human Growth & Development1.6.23DNA1.6.24Alleles1.6.25Genotype vs Phenotype1.6.26Punnett Squares1.6.27The Double Circulatory System1.6.28Heart and Blood1.6.29Blood Vessels1.6.30The Renal System1.6.31Glucose & Diabetes1.6.32Blood Glucose1.6.33Human Illnesses1.6.34Bacterial and Viral Infections1.6.35Antibiotics1.6.36Vaccinations1.6.37How Antibiotics and Vaccines Work1.6.38The Effects of Recreational Drug Use1.6.39Mental Health1.6.40Depression and Anxiety1.6.41End of Topic Test - Muscles & Bones1.6.42End of Topic Test - Respiration1.6.43End of Topic Test - DNA & The Circulatory System1.6.44End of Topic Test - Human Illnesses & Vaccination
2Chemistry
2.1Particles
2.1.1Particles2.1.2States of Matter2.1.3Compression of Substances2.1.4Shape of Substances2.1.5Changes of State2.1.6Elements and Atoms2.1.7Compounds2.1.8Diffusion2.1.9Changing State2.1.10Pressure2.1.11Temperature Increase in a Gas2.1.12Conservation of Mass2.1.13Purity of Substances2.1.14Pure Substances2.1.15Evaporation2.1.16Mixtures2.1.17Separating Mixtures2.1.18Distillation2.1.19Chromatography2.1.20Solubility2.1.21Investigating Solubility2.1.22Chemical Symbols2.1.23Chemical Formulae2.1.24Polymers2.1.25Ceramics2.1.26Composites2.1.27End of Topic Testing - Substances2.1.28End of Topic Testing - Mixtures2.1.29End of Topic Testing - Chemical Symbols & Polymers2.1.30Diagnostic Misconceptions - Particles2.1.31Diagnostic Misconceptions - Temperature2.1.32Diagnostic Misconceptions - Boiling Liquids
2.2Chemical Reactions
2.3Atoms, Elements, Compounds
2.4The Periodic Table
2.5Materials & the Earth
2.5.1The Composition of The Earth2.5.2The Structure of the Earth2.5.3Igneous Rocks2.5.4Sedimentary Rocks2.5.5Metamorphic Rocks2.5.6The Rock Cycle2.5.7Physical Weathering2.5.8Chemical Weathering2.5.9Biological Weathering2.5.10The Formation of Fossils2.5.11Crude Oil2.5.12The Earth's Early Atmosphere2.5.13The Earth's Atmosphere Today2.5.14Oxygen in the Atmosphere2.5.15Carbon Dioxide in the Atmosphere2.5.16Greenhouse Gases2.5.17Climate Change2.5.18Resources2.5.19Recycling2.5.20Ceramics2.5.21Polymers2.5.22Composites2.5.23End of Topic Test - Earth2.5.24End of Topic Test - Materials2.5.25End of Topic Test - Polymers2.5.26Diagnostic Misconceptions - Air2.5.27Diagnostic Misconceptions - Greenhouse Effect
2.6Reactivity
2.6.1Ions2.6.2Ionic Bonding2.6.3State Symbols2.6.4Balancing Chemical Equations2.6.5Relative Formula Mass2.6.6Calculating the Relative Formula Mass2.6.7The Reactivity Series2.6.8Carbon & The Reactivity Series2.6.9Displacement Reactions2.6.10Displacement Reactions - Halogens2.6.11Alloys2.6.12Metal Alloys2.6.13End of Topic Test - Ions2.6.14End of Topic Test - Reactivity
2.7Energetics
3Physics
3.1Energy
3.1.1Energy Stores & Pathways3.1.2Energy Transfers3.1.3Common Energy Transfers3.1.4Wasted Energy3.1.5Efficiency of Energy Transfer3.1.6Sankey Diagrams3.1.7Heat & Temperature3.1.8Heat Transfer3.1.9Conductors vs Insulators3.1.10Reducing Energy Transfers3.1.11Energy & Power3.1.12Energy in Food3.1.13Calories3.1.14Food Labels3.1.15Power Ratings of Appliances3.1.16Comparing Bulbs3.1.17Fuel Bills3.1.18Calculating Fuel Bills3.1.19Non-Renewable Energy - Fossil Fuels3.1.20Other Non-Renewables3.1.21Renewable Energy - Air & Ground3.1.22Renewable Energy - Water3.1.23Conservation of Energy3.1.24End of Topic Test - Energy3.1.25End of Topic Test - Energy & Non-Renewables3.1.26End of Topic Test - Energy in Things3.1.27Diagnostic Misconceptions - Stationary Objects
3.2Forces & Motion
3.2.1Forces3.2.2Contact Forces3.2.3Balanced Forces3.2.4Force Diagrams & Resultant Forces3.2.5Free Body Diagram - Uses3.2.6Force & Acceleration3.2.7Gravity3.2.8Weight3.2.9Pressure3.2.10Speed3.2.11Relative Motion3.2.12Friction 13.2.13Friction 23.2.14Turning Effect3.2.15Moment3.2.16Equation for Moments3.2.17Water & Air Resistance3.2.18Distance-Time Graphs3.2.19Levers3.2.20Work & Machines3.2.21Work3.2.22Machines3.2.23Elasticity3.2.24Elasticity - Hooke's Law3.2.25Archimedes Principle3.2.26Floating & Sinking3.2.27Vacuums3.2.28Thermal Energy & Conduction3.2.29Convection & Radiation3.2.30Evaporation3.2.31End of Topic Test - Forces3.2.32End of Topic Test - Motion3.2.33End of Topic Test - Work3.2.34End of Topic Test - Vacuums3.2.35Diagnostic Misconceptions - Weight vs Mass3.2.36Diagnostic Misconceptions - Gravity
3.3Waves
3.3.1Waves3.3.2Types of Waves3.3.3Observing Waves3.3.4Wave Speed3.3.5Earthquakes3.3.6Hearing3.3.7Observing Sound3.3.8Sound waves are Longitudinal Waves3.3.9Uses of Sound Waves3.3.10The Interactions of Sound with Different Mediums3.3.11Reflecting Sounds3.3.12The Speed of Sound3.3.13Measuring the Speed of Sound3.3.14The Hearing Range of Humans3.3.15The Human Ear3.3.16Light Waves3.3.17Coloured Light3.3.18Observing Light3.3.19Reflection3.3.20Drawing a Reflected Image3.3.21Refraction of Light 13.3.22Refraction of Light 23.3.23The Human Eye3.3.24The Eye as a Pinhole Camera3.3.25Lenses3.3.26Colour3.3.27Seeing Colour3.3.28Colours of Light3.3.29Drawing Waves3.3.30Wave Interactions3.3.31Comparing Sound & Light3.3.32General Principles of Radiation3.3.33Temperature & Black Bodies3.3.34Production of Electromagnetic Waves3.3.35Sound as Radiation3.3.36Light as Radiation3.3.37End of Topic Test - Sound3.3.38End of Topic Test - Light3.3.39End of Topic Test - Waves3.3.40End of Topic Test - Waves 23.3.41Diagnostic Misconceptions - Reflection3.3.42Diagnostic Misconceptions - Wave Speed
3.4Electricity & Magnetism
3.4.1Circuit Symbols3.4.2Resistors & Diodes3.4.3Electric Current3.4.4Measuring Current3.4.5Potential Difference3.4.6Series Circuits3.4.7Parallel Circuits3.4.8Resistance3.4.9Charges3.4.10Static Electricity3.4.11Magnets3.4.12Magnetic Fields3.4.13The Earth's Field3.4.14Electromagnetism3.4.15Uses of Electromagnets3.4.16Strength of Magnetic Fields3.4.17Circuit Symbols HyperLearning3.4.18End of Topic Test - Electricity3.4.19End of Topic Test - Magnetism3.4.20Diagnostic Misconceptions - Positive Charge3.4.21Diagnostic Misconceptions - Powering a Circuit
3.5Matter
3.5.1Physical Reactions3.5.2Changes of State3.5.3Particles3.5.4Density3.5.5Density & the Particle Model3.5.6The Equation for Density3.5.7Dissolving3.5.8Brownian Motion3.5.9Diffusion3.5.10Filtration3.5.11Solids3.5.12Liquids3.5.13Gases3.5.14Weight & Mass3.5.15Gravity3.5.16Gravitational Field Strength3.5.17Gravity in Space3.5.18Atmospheric Pressure3.5.19Liquid Pressure3.5.20End of Topic Test - States of Matter3.5.21End of Topic Test - Weight3.5.22Diagnostic Misconceptions - Floating
4Disciplinary Knowledge
4.1Thinking Scientifically
4.1.1Strengths & Limitations of Models4.1.2Symbols & Formulae to Represent Scientific Ideas4.1.3Analogies in Science4.1.4Changing Models – Atomic Theory4.1.5Working Safely in the Lab4.1.6Variables4.1.7Variables - Common Experiments4.1.8Writing a Hypothesis & Prediction4.1.9Planning an Experiment4.1.10Maths Skills for Science - Mean4.1.11Maths Skills for Science - Percentage & Equations4.1.12Drawing Scientific Apparatus4.1.13Observation & Measurement Skills4.1.14Types of Data4.1.15Graphs & Charts4.1.16Bias in Science4.1.17Conclude & Evaluate4.1.18End of Topic Test - Scientific Thinking4.1.19End of Topic Test - Experimenting
Practice questions on The pH Scale
Can you answer these? Test yourself with free interactive practice on Seneca — used by over 10 million students.
- 1Choose the alkaline substance:True / false
- 2Which of these is an acid?Multiple choice
- 3Which of these is an alkali?Multiple choice
- 4
- 5
Unlock your full potential with Seneca Premium
Unlimited access to 10,000+ open-ended exam questions
Mini-mock exams based on your study history
Unlock 800+ premium courses & e-books